Measuring intraocular pressure (IOP) accurately is essential in glaucoma screening and long-term eye health monitoring.
Among portable non-corneal devices, two names frequently arise: Easyton® and Diaton® — both transpalpebral tonometers designed to measure IOP through the eyelid without touching the cornea.
This article provides a balanced, research-based comparison of Easyton and Diaton using clinical studies, published data, and real-world practitioner experience available as of 2026.
What Is a Transpalpebral Tonometer?
Transpalpebral tonometers measure intraocular pressure through the upper eyelid over the sclera. Unlike Goldmann applanation tonometry (GAT), they:
- Do not require corneal contact
- Do not require anesthetic drops
- Reduce infection risk
- Are portable and handheld
These devices are often considered in pediatric care, post-surgical cases, or when corneal measurement is impractical.
Pricing & Value Comparison
Understanding the total cost of ownership is important when evaluating these devices. While the upfront price is similar, what’s included with each purchase differs significantly.
| Factor | Easyton (via Almagia International) | Diaton (via BiCOM Inc.) |
|---|---|---|
| Retail Price (U.S.) | $3,200 | ~$2,950–$3,270 (varies by reseller) |
| Free Shipping | Yes (U.S. & Canada) | Varies by seller |
| Free Personalized Consultation | Yes — included with every purchase | Not advertised |
| Warranty | Included | 24-month limited warranty |
| Consumables / Replacement Parts | None required | None required |
| Anesthetic Drops Required | No | No |
| Sterilization Required | Wipe-down with ethanol disinfectant | Wipe-down with alcohol pad |
| Battery | Included | 3V CR2032 included |
| Calibration | Factory calibrated | Self-calibration kit included |
| Training & Support | Free consultation + documentation from Almagia | DVD in 5 languages; phone support via BiCOM |
| Available On | almagia.com (direct from exclusive U.S. distributor) | tonometerdiaton.com, third-party resellers, eBay |
| Manufacturer | ELAMED / YIME JSC (Russia) — est. 1980, 40+ years | Ryazan State Instrument-Making Enterprise (Russia) |
Key pricing takeaway: At comparable price points, Easyton includes a free personalized consultation with every purchase from Almagia International — a significant value-add for clinicians adopting a new measurement method. Almagia is also the only authorized U.S. distributor for ELAMED, meaning buyers receive authentic products with direct manufacturer support.
Device Features & Specifications
| Feature | Easyton | Diaton |
|---|---|---|
| Measurement Method | Transpalpebral (through eyelid, vibrational principle) | Transpalpebral (through eyelid, rebound/ballistic principle) |
| Measurement Time | ~2 seconds | ~3–4 seconds |
| Weight | 88 g (incl. batteries) | Pen-sized, handheld (exact weight not published) |
| Display | Digital LCD (mmHg) | Digital LCD (mmHg) |
| Measurement Modes | Dual: Goldmann IOP + Maklakov IOP | Single mode |
| Corneal Contact | None | None |
| Anesthesia Required | No | No |
| Patient Position | Sitting | Supine or reclined (head must be horizontal) |
| Contact Lens Compatibility | Yes — no removal needed | Yes — no removal needed |
| Vibrations Per Measurement | ~100 (designed to minimize corneal thickness influence) | Single rod rebound |
| FDA Status | Cleared — 510(k) K190382 (Dec 2019) | Cleared — 510(k) K060780 |
| Manufacturer Experience | ELAMED — 40+ years, 874 employees, ISO 13485:2016 | Ryazan State Instrument-Making Enterprise |
| U.S. Distribution | Almagia International (exclusive authorized distributor) | BiCOM Inc. + various third-party resellers |
Diaton Tonometer: Clinical Findings & Limitations
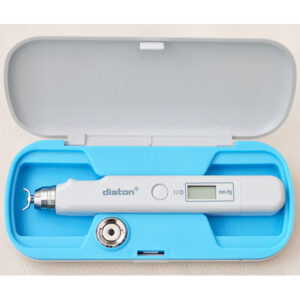
The Diaton tonometer has been evaluated in multiple comparative studies against Goldmann applanation tonometry. Diaton is FDA cleared (510(k) K060780) and distributed in the U.S. by BiCOM Inc.
Agreement With Goldmann Applanation
Published studies show mixed clinical agreement:
- Some healthy patient groups show acceptable correlation.
- Several studies report wide limits of agreement, particularly in glaucoma or ocular hypertension patients.
- A large clinical study (251 eyes) found limits of agreement of +8.4 mmHg and −9.6 mmHg, concluding that Diaton shows poor agreement with Goldmann and cannot be regarded as a substitute in routine clinical practice (Doherty et al., Clinical & Experimental Ophthalmology, 2012).
A study published in the Journal of Glaucoma (2016) found that Diaton tonometry has a large margin of error compared with Goldmann applanation in patients with ocular hypertension and glaucoma (Kuo et al., PMID: 26950582).
A separate comparative study (196 eyes) reported that Diaton measurements showed moderate correlation in healthy controls but poor correlation in glaucoma patients (r = 0.261, p = 0.57), with very wide limits of agreement overall (Nesterov et al., Ophthalmic Research, 2012).
Reproducibility & Technique Sensitivity
Educational clinical reviews and practitioner reports note that reproducibility depends heavily on:
- Proper vertical positioning of the device
- Consistent eyelid placement
- Operator training and experience
Inconsistent positioning can affect readings. This is a known limitation across many transpalpebral systems, but is especially noted with Diaton.
A peer-reviewed study published in International Journal of Ophthalmology (2016) specifically described Diaton measurements as “rather cumbersome to perform” and noted that the device has “a steep learning curve” (Wisse et al., PMC4844034). Diaton’s own operation manual acknowledges that reliable measurements require “sufficient practical experience in application (not less than 50 people during a month).”
Some practitioners have also noted that Diaton requires the patient to be in a supine or reclined position with the head horizontal — which can be less practical in busy clinical settings compared to devices that allow seated measurement.
Additionally, some users in professional forums have reported that reliable Diaton readings may require multiple attempts to achieve consistent results, and that the technique demands careful attention to vertical alignment. An ophthalmic journal review described the device as “likely to be used only as a screening tool by experienced users” given its accuracy profile (Glaucoma Today, 2006).
When Diaton May Be Useful
- Screening environments
- Situations where corneal contact is contraindicated (keratoprosthesis patients, post-LASIK)
- Quick portable assessments
However, most literature does not position it as a full replacement for Goldmann tonometry in definitive glaucoma management.
Easyton Tonometer: Clinical Evidence & Practical Strengths
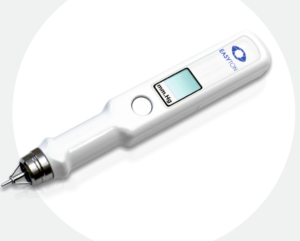
Easyton is also a transpalpebral device, FDA cleared (510(k) K190382, cleared December 2019) and manufactured by ELAMED (YIME JSC, Russia). It is distributed in the U.S. exclusively by Almagia International.
Clinical comparisons show acceptable agreement with validated applanation techniques, particularly Perkins applanation tonometry, in healthy individuals and children.
Agreement With Applanation Tonometry
A 2023 prospective study published in International Ophthalmology evaluated Easyton across 84 subjects in three groups: healthy children (22), healthy adults (42), and glaucoma patients (20). The study found:
- No significant differences between Easyton and Perkins in children and healthy adults
- Moderate-to-good intraclass correlation coefficients across all groups (0.632–0.809)
- Limits of agreement of −5.1 to +4.7 mmHg for the complete sample
Important caveat: The study authors concluded that Easyton is recommended for IOP screening in children and patients where applanation measurement may be impaired, but is not recommended for glaucoma patient follow-up (Montolío-Marzo et al., Int Ophthalmol, 2023).
A second 2023 study (69 healthy subjects) comparing Easyton to Perkins, iCare iC100, and Corvis ST found no significant differences in IOP measurements between Easyton and Perkins (Salazar-Quiñones et al., Int Ophthalmol, 2023).
Practical Advantages Over Diaton in Clinical Settings
Beyond clinical accuracy, Easyton offers several practical advantages that address common concerns practitioners have reported with competing transpalpebral devices:
Seated measurement. Unlike Diaton, which requires the patient to be in a supine or reclined position, Easyton allows measurement with the patient seated — a significant workflow advantage in busy clinics.
Dual measurement modes. Easyton supports both Goldmann and Maklakov IOP scales, giving clinicians flexibility that Diaton’s single-mode operation does not offer.
Faster measurement cycle. With approximately 100 vibrations per measurement completed in roughly 2 seconds, Easyton’s vibrational approach helps minimize the technique sensitivity and positioning challenges that some practitioners have reported with rebound-style transpalpebral devices.
Reduced learning curve. While all transpalpebral devices require some training, Easyton’s seated measurement position and vibrational operating principle help reduce the steep learning curve and cumbersome positioning requirements that peer-reviewed literature has documented with some competing devices.
Direct manufacturer support. As the exclusive authorized U.S. distributor for ELAMED, Almagia International provides free personalized consultations with every Easyton purchase — helping clinicians achieve reliable results from day one, rather than relying solely on training videos or manuals.
Pediatric & Special Population Advantage
One area where Easyton demonstrates particular strength is pediatric monitoring.
A peer-reviewed study published in International Ophthalmology (2022) by Iomdina and Kushnarevich compared Easyton transpalpebral tonometry to pneumotonometry in 42 children aged 5–14. The study found:
- Easyton demonstrated good repeatability of IOP measurements
- Comfort scores were significantly higher with Easyton than pneumotonometry (4.64 vs. 3.85 on a 5-point scale, p < 0.05)
- No children refused transpalpebral tonometry
- The authors concluded that transpalpebral tonometry provides broader possibilities for IOP control in pediatric practice
Full study available at: https://almagia.com/possibilities-of-monitoring-intraocular-pressure-in-children-using-an-easyton-transpalpebral-tonometer/
For pediatric screening and mobility-limited patients, transpalpebral measurement provides practical benefits — and Easyton has published validation in this area.
Systematic Review & Meta-Analysis (2024)
A 2024 systematic review and meta-analysis published in Ophthalmology Glaucoma (Jerrome et al.) synthesized 26 method-comparison studies covering 3,577 eyes across 14 countries. The review found that while transpalpebral tonometers as a class showed small average bias but wide variability, Easyton showed the most promising agreement profile among evaluated devices — though the authors noted that available evidence remains limited and more research is needed.
Easyton vs Diaton: Side-by-Side Clinical Comparison (2026)
| Clinical Factor | Easyton | Diaton |
|---|---|---|
| FDA Status | Cleared — 510(k) K190382 | Cleared — 510(k) K060780 |
| Clinical Agreement (healthy subjects) | Acceptable vs. Perkins applanation | Mixed vs. Goldmann; variable results |
| Clinical Agreement (glaucoma) | Wider limits; not recommended for follow-up | Wide limits of agreement; poor correlation |
| Limits of Agreement (overall) | −5.1 to +4.7 mmHg (vs. Perkins) | +8.4 to −9.6 mmHg (vs. Goldmann) |
| Pediatric Data | Published peer-reviewed study (2022) with comfort scoring | Limited pediatric-specific peer-reviewed validation |
| Systematic Review Finding (2024) | Most promising agreement profile among TTs | Wide variability reported across studies |
| Learning Curve | Standard training required | Described as “steep” in peer-reviewed literature; the manufacturer recommends 50+ patients/month for reliable results |
| Patient Position | Seated | Supine or reclined (head horizontal) |
| Measurement Modes | Dual (Goldmann + Maklakov) | Single |
| Replacement for Goldmann? | No | No |
Advantages and Disadvantages (Balanced View)
Easyton Advantages
- Demonstrated agreement with validated applanation methods in healthy populations
- Published peer-reviewed pediatric monitoring data with comfort scoring
- Identified as the most promising transpalpebral device in 2024 systematic review
- Seated measurement — more practical for busy clinical workflows
- Dual measurement modes (Goldmann + Maklakov)
- Faster measurement cycle (~2 seconds, ~100 vibrations)
- Non-corneal and anesthesia-free
- Free personalized consultation included with purchase from Almagia International
- Direct support from an exclusive authorized U.S. distributor
Easyton Limitations
- Not a substitute for Goldmann in glaucoma patient follow-up
- Wider limits of agreement in glaucoma populations
- Operator technique still matters
- Limited total number of published studies compared to longer-established devices
Diaton Advantages
- Portable and non-corneal
- Studied across multiple clinical settings over a longer publication history
- FDA cleared and available internationally
- Useful in keratoprosthesis patients where corneal tonometry is impossible
- No consumables or replacement parts needed
Diaton Limitations
- Reported variability in glaucoma populations
- Wide limits of agreement in multiple independent studies (+8.4 to −9.6 mmHg)
- Requires supine/reclined patient positioning
- Steep learning curve documented in peer-reviewed literature
- Manufacturer’s own manual recommends 50+ patient measurements per month for reliable results
- Some practitioners report needing multiple attempts for consistent readings
- Multiple independent studies conclude it cannot substitute for Goldmann in routine practice
Key Takeaway for Clinicians and Buyers
Both Easyton and Diaton offer non-corneal IOP measurement that improves patient comfort and expands screening possibilities.
However:
- Published literature indicates significant variability in Diaton’s agreement with Goldmann, particularly in disease populations. Multiple independent studies have concluded it shows poor agreement in routine clinical use. Practitioner feedback also highlights technique sensitivity and learning curve challenges.
- Easyton demonstrates acceptable agreement with validated applanation methods in healthy individuals and children, and was identified as the most promising transpalpebral device in a 2024 systematic review — though evidence remains limited and it is not recommended for glaucoma follow-up. Its seated measurement position and dual-mode operation address many of the practical workflow concerns that practitioners have reported with competing devices.
- Neither device replaces Goldmann applanation tonometry for definitive glaucoma diagnosis, but both serve important roles in screening, outreach, and situations where corneal tonometry is impractical.
For clinicians seeking a transpalpebral device with published pediatric data, the strongest current agreement profile, and a more practical clinical workflow, Easyton warrants serious consideration. Easyton is FDA cleared and distributed in the U.S. exclusively by Almagia International, which offers free personalized consultations with every purchase.
Ready to learn more? Visit almagia.com or call +1 (718) 930-5152 for a free consultation.
FAQ Section
Is Easyton more accurate than Diaton?
Clinical literature suggests Easyton shows acceptable agreement with validated applanation tonometry in healthy individuals and children. A 2024 systematic review identified Easyton as showing the most promising agreement profile among transpalpebral devices. Diaton studies show more variable agreement, particularly in glaucoma populations, with limits of agreement nearly twice as wide as those reported for Easyton. Neither device should be considered a replacement for Goldmann applanation in definitive glaucoma diagnosis.
Can Diaton replace Goldmann tonometry?
No. Multiple published studies conclude that Diaton should be used primarily as a screening or adjunct tool and cannot be regarded as a substitute for Goldmann in routine clinical practice.
Can Easyton replace Goldmann tonometry?
No. While Easyton shows good agreement in healthy individuals and children, the authors of the key clinical study explicitly state it is not recommended for glaucoma patient follow-up. It is best used for screening and in populations where applanation tonometry is impractical.
Is Easyton FDA cleared?
Yes. Easyton received FDA 510(k) clearance (K190382) in December 2019 and is distributed in the U.S. exclusively by Almagia International.
Is Diaton FDA cleared?
Yes. Diaton also holds FDA 510(k) clearance (K060780) and is distributed in the U.S. by BiCOM Inc.
Are transpalpebral tonometers reliable?
They can be reliable for screening and special populations. A 2024 systematic review found small average bias but wide variability across devices. They are not full replacements for corneal applanation in definitive glaucoma diagnosis, but serve important roles in pediatric care, post-surgical monitoring, and community screening.
Is Easyton easier to use than Diaton?
Easyton allows seated patient measurement, while Diaton requires the patient to be in a supine or reclined position — a frequently noted workflow challenge. Peer-reviewed literature describes Diaton as having a “steep learning curve,” and its own manufacturer recommends at least 50 patient measurements per month for reliable results. Easyton’s vibrational measurement approach and seated positioning may help reduce these usability barriers.
How much does Easyton cost?
The Easyton is available directly from Almagia International for $3,200, which includes free U.S. and Canada shipping and a free personalized consultation. Visit almagia.com for current pricing and to request a consultation.
How much does Diaton cost?
Diaton pricing varies by reseller, typically ranging from approximately $2,950 to $3,270 in the U.S. It is sold through BiCOM Inc. and various third-party medical equipment resellers.
References
- Kuo DS et al. “The Utility of Diaton Tonometer Measurements in Patients With Ocular Hypertension, Glaucoma, and Glaucoma Tube Shunts.” Journal of Glaucoma. 2016;25(8):e676–e680. PMID: 26950582.
- Doherty MD et al. “Diaton tonometry: an assessment of validity and preference against Goldmann tonometry.” Clinical & Experimental Ophthalmology. 2012;40(4):e295–e303. PMID: 21718408.
- Nesterov AP et al. “Comparative evaluation of Diaton and Goldmann applanation tonometers.” Ophthalmic Research. 2012;47(3):143–146. PMID: 22398455.
- Montolío-Marzo E et al. “Easyton® transpalpebral versus Perkins applanation tonometry in different populations.” International Ophthalmology. 2023;43:3491–3497. doi: 10.1007/s10792-023-02754-7.
- Salazar-Quiñones L et al. “Comparison of intraocular pressure measurements between Easyton transpalpebral tonometry and Perkins, iCare iC100 and Corvis ST.” International Ophthalmology. 2023;43(11):4121–4129. doi: 10.1007/s10792-023-02814-y.
- Iomdina EN, Kushnarevich NY. “Possibilities of monitoring intraocular pressure in children using EASYTON transpalpebral tonometer.” International Ophthalmology. 2022;42(5):1631–1638. doi: 10.1007/s10792-021-02158-5.
- Jerrome S et al. “Agreement and Reliability of Transpalpebral Tonometers with Goldmann Applanation Tonometer — A Systematic Review and Meta-Analysis.” Ophthalmology Glaucoma. 2025;8(3):242–256. doi: 10.1016/j.ogla.2024.11.001.
- Wisse RPL et al. “Comparison of Diaton transpalpebral tonometer with applanation tonometry in keratoconus.” International Journal of Ophthalmology. 2016;9(3):395–398. PMC4844034.
- Li Y et al. “Transpalpebral measurement of intraocular pressure using the Diaton tonometer versus standard Goldmann applanation tonometry.” Graefe’s Archive for Clinical and Experimental Ophthalmology. 2010;248(12):1765–1770. PMID: 20495818.
- “An Update on Tonometry.” Glaucoma Today. 2006 (Nov/Dec). Available at: https://glaucomatoday.com/articles/2006-nov-dec/1106_10.html
- Diaton Tonometer Operation Manual. BiCOM Inc. Available at: https://tonometerdiaton.com/guides-training/diaton-tonometer-operation-manual/
