PRODUCT DESCRIPTION
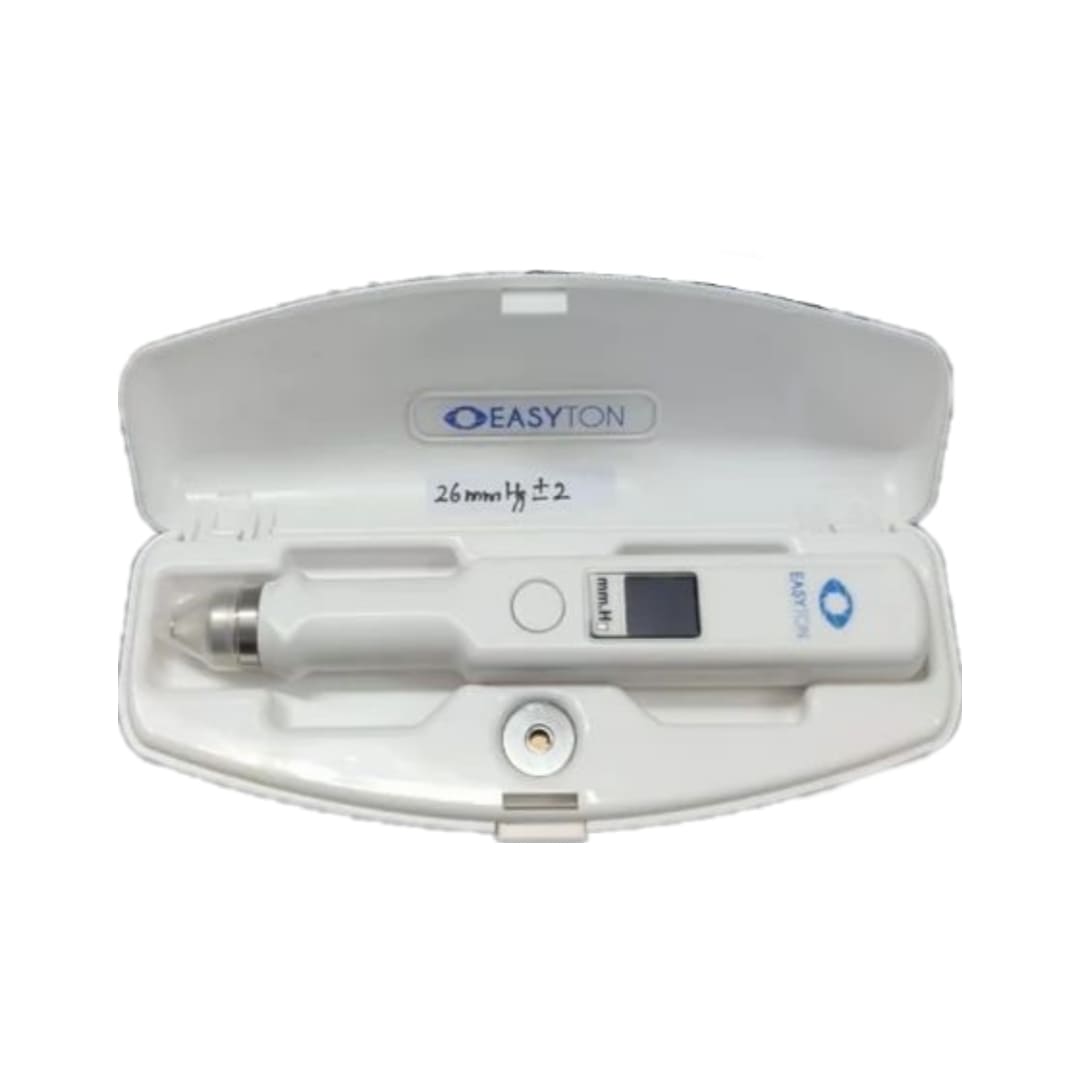
IOP Tonometer (applanation tonometry) is a unique blend of technology and science. By its very nature, it is a tonometer eye pressure measurement device that relies on the transpalpebral method to provide as accurate results as those of intracameral IOP tonometry. This Pen Tonometer is designed for ophthalmologists to be used in healthcare settings to encourage improvement in the diagnosis, monitoring, and treatment of glaucoma and other ocular conditions.
This Easyton Tonometer (pen tonometer) is approved by FDA. PDF
The effectiveness is backed up by many accuracy, reproducibility, and repeatability tests. This Easyton tonometer has also been proven to deliver next-level diagnostics performance in the study with the Goldmann method as a reference standard. We take pride in offering this eye Easyton tonometer for sale to help eye care specialists feel the difference in IOP measurement. It can be done easier with the guaranteed accuracy when this tonometer is at hand.
Operating Principle of the Applanation Tonometry for Eye Pressure Measurement
The functionality of our device (applanation tonometry) and the way it works cater to the needs of ophthalmologists as well as patients. Designed to simplify the process of eye pressure diagnostics and make it less stressful for people, the device is based on the vibrational operating principle. Tonometer or IOP tonometer helps minimize fluctuations and irregularities that often come with other IOP measurement methods.
Once the tonometer (applanation tonometry) for eye pressure diagnostics is applied, it starts producing multiple vibrations at different frequency levels (depending on how high or low the measured IOP is). The process begins as soon as you turn on the device and place the rod on the patient’s eyelid. This Easyton tonometer, a device then automatically processes pressure readings and recalculates them into millimeters of mercury (mmHg) so that you can see the average IOP value on its display screen.
Our tonometer eye pressure device (applanation tonometry) produces approximately 100 vibrations with a single push of the rod. Most of them are aimed at preventing the corneal thickness factor from affecting the results. By making up for the possible ambiguity of measurements, these vibrations are pivotal to ensure pinpoint accuracy of the IOP value.