Abstract
Objective: To compare the effects of low-intensity pulsed ultrasound (LIPUS) or pulsed electromagnetic fields (PEMF) on fracture healing through a systematic review of original, English-language clinical research reports.
Data Sources: A search of MEDLINE, Physiotherapy Evidence Database (PEDro), and ProQuest to identify clinical trials of LIPUS or PEMF with fractures in humans, written in English, published from 1966 through 2004. Key words were ultrasound, fracture, tibial, electric current, and healing.
Study Selection: After search limits were applied, 17 papers were assessed independently by 2 reviewers. Papers were excluded from consideration if they lacked (1) random allocation of treatments, (2) inclusion of skeletally mature patients of either sex with a current fracture, (3) blinding of both the patient and the assessors as to treatment group, (4) administration of either LIPUS or PEMF treatments to one of the treatment groups, or (5) assessment of time to fracture healing or proportion of fractures healed, as determined radiographically, clinically, or both.
Data Extraction: Eight trials met the inclusion criteria. Methodologic quality of all trials was assessed using the PEDro criteria. Outcome measures were tabulated.
Data Synthesis: Heterogeneity among studies precluded direct comparison of the efficacy of LIPUS to that of PEMF.
Conclusions: The studies we included in our review were of generally high methodologic quality. The evidence suggests that LIPUS may speed healing of acute tibial fractures. Comparison studies of these modalities are needed to guide treatment of fractures sustained by athletic individuals.
Low-intensity pulsed ultrasound (LIPUS) and pulsed electromagnetic fields (PEMF) are used in fracture care. What is the effect of such interventions on the acceleration of acute fracture healing and the healing of nonunions? Do treatment responses to these devices differ?
We have observed an increase in the number of athletes receiving LIPUS or PEMF treatments to promote fracture healing. Numerous authors1–16 have reported that LIPUS or PEMF with specific energy delivery settings have positive effects in bone healing. The bones most frequently mentioned in the research are the vertebrae and long bones.
We also have encountered questions from students and athletes as to which of the devices is more effective. In response, we searched for comparison studies. Finding none, we conducted a systematic review of clinical trials concerning the treatment of fractures with LIPUS and PEMF bone stimulators in order to assess and compare the effects on fracture healing time. We believe that all providers involved in the care of injured athletic patients should have an understanding of each aspect of the plan of care and should be able to address or appropriately direct questions from the athlete-patient. Our report provides current responses to these questions and provides direction for future investigations that will offer more definitive answers.
METHODS
Two authors (N.A.W. and J.P.) independently identified relevant randomized clinical trials through a systematic search of several electronic databases: MEDLINE (1966 through 2004), Physiotherapy Evidence Database (PEDro) (1929 through 2004), and ProQuest (1987 through 2004) (Figure). The key words we used were ultrasound, fracture, and tibial with limits of human, clinical trials, and written in English. We initially focused on the tibia because a cursory review of the literature suggested that this bone would allow for the best opportunity to compare responses to the devices. However, this search yielded only 4 papers. Therefore, we removed the limits from the search criteria and found 5 additional studies. The authors of 4 of the studies used ultrasound for imaging; authors of the fifth provided a cost analysis.
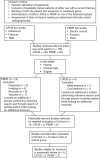
To find additional studies of LIPUS, we performed a hand search of the references within the clinical reports and added 2 papers. We conducted a similar search using the key words electric current, fracture, and tibialin addition to the previously mentioned limits. This search yielded 2 papers. In an attempt to locate additional research, we dropped the limits and found 13 studies, but of these, 2 were diagnostic in nature, 3 related to imaging, 2 involved muscular healing, and 1 addressed vascular response. Thus, a total of 5 clinical trials of PEMF for fracture healing remained for further investigation.
Due to the small number of trials relating to tibial fracture healing that were identified, we expanded our search to include treatment of fractures in all bones with LIPUS or PEMF. First, we searched using the aforementioned limits with ultrasound, fracture, and bone. This yielded 58 papers. The key word healingwas added to the list, resulting in 7 studies. Four involved only treatment of tibial fractures, 1 was an imaging study, another was specific to diagnostics, and 1 reported on healing of the femur, radius, ulna, humerus, metatarsal, and clavicle with LIPUS.8 A similar search was then done for electric current, in place ofultrasound in the previous search. This yielded 3 papers, 2 of which concentrated on the tibia only, and 1 related to the femur, tibia, radius, and humerus.14 These additional studies were retained for this review.
We independently applied previously established inclusion criteria to assess the methods of each potentially eligible study. The following criteria had to be met: random allocation of treatments, inclusion of skeletally mature patients of either sex with a current fracture, blinding of both the patient and the assessors as to treatment group, administration of either LIPUS or PEMF treatments to one of the treatment groups, and assessment of time to fracture healing or proportion of fractures healed as determined radiographically, clinically, or both.
We assessed the quality of the selected studies and independently abstracted the data. For a checklist, we adopted the PEDro 10-point scale.17 The PEDro scale is based on 2 premises, internal validity and adequate statistical information to allow for interpretation.17 In a subsequent consensus meeting, we discussed each criterion. Because the articles already were rated in PEDro, the ratings were used as a third voice in the assessment process. When we disagreed about assessments, we discussed our concerns until we attained a consensus.
RESULTS
We identified 159 studies related to LIPUS (135) and PEMF (24), of which 14 were human clinical trials published in English (Figure). Of the 14 studies, 2 were excluded from the final analysis because another form of electric stimulation was studied.12,13 Four were excluded8,10,11,14 due to lack of a control group for comparison (Table 1). The PEDro scores for the 8 retained and 6 excluded studies are found in Table 1.

Study Characteristics
We extracted and tabulated the study population, intervention, outcomes, and follow-ups; we reported results of the assessed trials. The 8 trials that were retained for analysis involved 321 patients, although Emami et al4,5 reported on the same patients in both of their papers; thus, 291 different patients were included. The sample sizes ranged from 16 to 67, and the mean number of patients studied per investigation was 40.1. The most frequent mechanism of injury for a fracture of the tibia was a motor vehicle accident. Authors of 4 studies8,10,12,14 included assessment of fracture healing of the femur, humerus, radius/ulna, fibula, fifth metatarsal, talus, scaphoid, and clavicle; however, none of these trials included a control group.
On assessing the other characteristics of the included studies, we noted considerable heterogeneity. The clinical trials were heterogeneous with regard to subject demographics, management of fractures in terms of casting and fixation, measurements of healing, timing of assessments, and the specific outcomes measured.
Outcome Measures
Radiographic healing of 3 of the 4 cortices was the most common outcome measure3–6,15,16 and is considered the gold standard. Some investigators, however, reported the number of days to healing,3–7,17,18whereas others reported the proportion of healed fractures at specific time points.9,15,16 Clinical analyses of healing provided another research measure. Included in the clinical analyses were pain via visual analog scale,9,15 tenderness via palpation and weight-bearing status,7,10 and degrees of motion at the fracture site upon physical stress.10,15 Most of the authors recorded a final healing time and lacked continuous measures over time.
Effectiveness of Treatment
The 8 reports3–7,9,15,16 included in our final assessment are summarized in Tables 2 and


Summary of Clinical Reports: Low-Intensity Pulsed Ultrasound Versus Placebo Low-Intensity Pulsed Ultrasound
Five sets of authors3–7 compared the healing rates of acute tibia fractures treated with active LIPUS or placebo LIPUS. Fracture management differed between the studies; details and sample sizes for each are reported in Table 4. The treatment values for these 5 studies3–7 were a burst width of 200 microseconds containing 1.5-MHz sine waves, with a repetition rate of 1 KHz and a spatial average temporal intensity of 30 mV/ cm2. In each study, LIPUS was applied 20 minutes daily. The total duration of treatment differed, however, and ranged from 75 days4,5 to 140 days,3,6 with Leung et al7 treating for 120 days. The inclusion and exclusion criteria of the 5 studies3–7 were similar. Emami et al4,5 treated fractures with a statically locked intramedullary rod, whereas Cook et al3 and Heckman et al6 treated fractures with closed reduction and cast immobilization. Patients in the Leung et al7 trial experienced complex fractures requiring internal or external fixation. It is unclear if differences in fracture management influenced the healing response to LIPUS.

Emami et al4,5 reported nonsignificant differences in radiographic union rate (125 days for placebo, 155 days for active treatment), orthopaedic clinical assessment of fracture healing (125 days for placebo, 129 days for active treatment), and the average time from fracture until radiographic evidence of the first callus formation (37 days for placebo, 40 days for active treatment).
Cook et al,3 Heckman et al,6 and Leung et al7 reported faster radiographic and clinical healing in those patients treated with active LIPUS. Cook et al3 specifically focused on the effects of the active versus placebo LIPUS treatments on smokers and nonsmokers. Cook et al3 showed reductions in time to clinical and radiographic healing in nonsmokers treated with LIPUS (84 and 96 days, respectively) versus the placebo control (96 and 129 days, respectively). They also found differences in the time to clinical and radiographic healing in smokers treated with LIPUS (103 and 96 days, respectively) versus the placebo control (175 and 125 days, respectively). The differences in time to healing reported in each of the studies3,6,7 were statistically significant.
Summary of Clinical Reports: Pulsed Electromagnetic Field Versus Placebo Pulsed Electromagnetic Field
Three groups9,15,16 compared active treatments of PEMF with placebo treatments of PEMF; however, treatment settings in each study were different. Barker et al9 used active machines producing a 1- to 5-mT peak, 5-millisecond burst waveform repeated at 15 Hz for 12 to 16 hours a day. Sharrard’s15 signal consisted of bursts of 20 individual pulses of a quasi-rectangular form followed by a sharper reverse form, with the bursts repeated at 15 Hz for 12 hours a day. Finally, Simonis et al16 used a peak current of 6 A at 150 V with the duration of the generated pulse set at 3 milliseconds in intervals of 40 milliseconds for 14 hours a day. Variations in treatment settings and the energy delivered to the tissues may have led to differences in outcomes. We were unable to locate studies comparing PEMF settings.
Sharrard15 and Simonis et al16 reported that a greater proportion of patients demonstrated radiographic healing when treated with PEMF than with placebo at 12 and 15 weeks, respectively. Patients in the Simonis et al16 study were treated after an oblique fibular osteotomy and application of a unilateral fixator. Those in the Sharrard15 study were treated with a full-leg plaster cast and restricted from weight bearing. Barker et al9defined a healed fracture when 2 independent observers were unable to detect movement at the fracture site with stress and with lack of a gap on radiograph. Barker et al9 found little difference in the rate of nonunion at 24 weeks in patients treated with and without PEMF who also received immobilization with a full-leg plaster cast and were restricted from weight bearing. Sharrard15 found that less tenderness was experienced by patients treated with PEMF (1.6) than with placebo (2.7) when assessed on a visual analog scale rating at 12 weeks posttreatment (P = .18).
DISCUSSION
Low-intensity pulsed ultrasound and PEMF are used to treat fractures of all types. However, a relatively limited number of randomized, controlled clinical trials have been used to investigate the efficacy of these treatments. Focusing on tibial fractures, we found 6 studies suggesting a benefit to using LIPUS and PEMF for fracture union.3,6,7,9,15,16 Others, however, have failed to show a beneficial effect.4,5 The studies we included in our review were of generally high methodologic quality (rating 8 or higher on the PEDro scale). To our knowledge, ours is the first systematic review attempting to compare the efficacy of LIPUS with that of PEMF. Unfortunately, such a comparison is not possible due to the heterogeneity in the outcomes measured and the timing of assessments. Moreover, the PEMF studies involved treatment of nonunions (Table 4), whereas the studies of LIPUS involved treatment of acute fractures.
The difference in the findings of Emami et al4,5 and the other LIPUS investigators3,6,7 could be the result of other factors, including management. The descriptions of the natures of the fractures were similar across studies (Table 4), however, except that those in the Leung et al7 report sustained more complex fractures. The patients included in the reports by Emami et al4,5 were all treated with intramedullary nails. The effect of intramedullary nailing on responses to LIPUS has not, to our knowledge, been reported.
Other than the reports by Emami et al,4,5 the papers3,6,7 we included in our review are in agreement with a previous meta-analysis by Heckman et al.1 The authors1 concluded that LIPUS may reduce the time to fracture healing for fractures treated nonoperatively. Effect sizes were calculated to estimate the magnitude of the treatment effect.20 Cohen19proposed that effect sizes of 0.2 represent small differences; 0.5, moderate differences; and 0.8+, large differences. Effect sizes for radiographic and clinical healing ranged from 1.9 magnitude7 to 4.2 magnitude,6 whereas reports of clinical healing alone ranged from 0.5 magnitude7 to 2.7 magnitude.6
The meta-analysis by Heckman et al1 and the studies we included investigated healing of tibial fractures. Kristiansen et al21 reported accelerated healing (38% less time to union than in controls) of dorsally angulated radial fractures in a multicenter, prospective, controlled clinical trial. We found no randomized, controlled clinical trials involving LIPUS in the treatment of fracture nonunions.
We limited our search to clinical trials that included a control group and were written in English. In 2002, LIPUS was approved by the Food and Drug Administration for the treatment of established nonunions.22The use of patients as their own controls in studies investigating the treatment of nonunion fractures is now accepted research practice. Nolte et al8 reported that 80% of the femur, radius, and scaphoid fractures treated with LIPUS healed in an average of 155 days and 75% of “other” bones healed in approximately 167 days, despite a long period of nonunion.
Authors of each of the randomized, controlled trials related to healing of nonunion fractures with PEMF, however, reported cases of healing with placebo stimulation. The lack of control groups precludes conclusions that no healing would have occurred without intervention. Yet the high success rates reported by Nolte et al8 certainly suggests that LIPUS is effective in the treatment of delayed healing and nonunion fractures.
Few high-quality controlled clinical trials of PEMF exist. We found 2 reports of PEMF fracture treatments that did not involve a control group, in addition to those we included. Each group reported high rates of long-bone union between 14 weeks and 5.2 months. Regarding long-bone fractures, Satter Syed et al14 found that 84.6% (11 of 13) experienced successful bone healing within an average treatment period of 14 weeks. Similarly, when Bassett et al10 studied the effects of PEMF on the tibia and the femur, the healing rate was 93% (65 of 70) or greater in an average of 4 months. Bassett et al10 performed additional work with the tibia and once again found that, in an average of 5.2 months, 87% (110 of 127) of all nonunions had healed with non–weight-bearing treatment. In addition to the results of the randomized clinical trials, the findings of Satter Syed et al14 and Bassett et al10suggest that PEMF speeds healing of tibial and other long-bone fractures.
CONCLUSIONS
Because the authors of the controlled LIPUS trials reported days to healing and those studying PEMF provided proportions of the groups with united fractures, it is not possible to compare the effects of these interventions. We recommend that investigators in future PEMF studies record time to healing in days, similar to what is done in LIPUS research.
Low-intensity pulsed ultrasound speeds acute fracture healing and promotes healing in nonunion fractures. At present, the efficacy of this treatment is best supported by the randomized, controlled clinical trials of acute tibial fractures3,14,15 available. Controlled clinical trials assessing the management of other acute fractures are needed. Evidence suggests that PEMF also increases the proportion of nonunion fractures that heal without additional intervention.
Comparison studies of these 2 modalities may prove useful in helping clinicians to select the most effective treatments for various fractures across a spectrum of patients. We also recommend that investigators stratify patients based on fracture status (acute versus nonunion), fixation (internal versus external), and smoking. Smoking tobacco slows down the regeneration of bone tissue,7 and a disproportionate number of smokers in either a treatment or a control group may bias the results in favor or disfavor of fracture treatments.
REFERENCES
- Heckman JD, Sarasohn-Kahn J. The economics of treating tibia fractures: the cost of delayed unions. Bull Hosp Joint Dis. 1997;56:63–72. [PubMed]
- Busse JW, Bhandari M, Kulkarni AV, Tunks E. The effect of low-intensity pulsed ultrasound therapy on time to fracture healing: a meta-analysis. Can Med Assoc J. 2002;166:437–441.[PMC free article] [PubMed]
- Cook SD, Ryaby JP, McCabe J, Frey JJ, Heckman JD, Kristiansen TK. Acceleration of tibia and distal radius fracture healing in patients who smoke. Clin Orthop Relat Res. 1997;337:198–207.[PubMed]
- Emami A, Petren-Mallmin M, Larsson S. No effect of low-intensity ultrasound on healing time of intramedullary fixed tibial fractures. J Orthop Trauma. 1999;13:252–257. [PubMed]
- Emami A, Larsson A, Petren-Mallmin M, Larsson S. Serum bone markers after intramedullary fixed tibial fractures. Clin Orthop Relat Res. 1999;368:220–229. [PubMed]
- Heckman JD, Ryaby JP, McCabe J, Frey JJ, Kilcoyne RF. Acceleration of tibial fracture-healing by non-invasive, low-intensity pulsed ultrasound. J Bone Joint Surg Am. 1994;76:26–34. [PubMed]
- Leung KS, Lee WS, Tsui HF, Liu PP, Cheung WH. Complex tibial fracture outcomes following treatment with low-intensity pulsed ultrasound. Ultrasound Med Biol. 2004;30:389–395. [PubMed]
- Nolte PA, van der Krans A, Patka P, Janssen IM, Ryaby JP, Albers GH. Low-intensity pulsed ultrasound in the treatment of nonunions. J Trauma. 2001;51:693–702. [PubMed]
- Barker AT, Dixon RA, Sharrard WJ, Sutcliffe ML. Pulsed magnetic field therapy for tibial non-union. Interim results of a double-blind trial. Lancet. 1984;994–996. [PubMed]
- Bassett CA, Mitchell SN, Schink MM. Treatment of therapeutically resistant non-unions with bone grafts and pulsing electromagnetic fields. J Bone Joint Surg Am. 1982;64:1214–1220. [PubMed]
- Bassett CA, Mitchell SN, Gaston SR. Treatment of ununited tibial diaphyseal fractures with pulsing electromagnetic fields. J Bone Joint Surg Am. 1981;63:511–523. [PubMed]
- Benazzo F, Mosconi M, Beccarisi G, Galli U. Use of capacitive coupled electric fields in stress fractures in athletes. Clin Orthop Relat Res. 1995;310:145–149. [PubMed]
- Brighton CT, Shaman P, Heppenstall RB, Esterhai JL, Jr, Pollack SR, Friedenberg ZB. Tibial nonunion treated with direct current, capacitive coupling, or bone graft. Clin Orthop Relat Res.1995;321:223–234. [PubMed]
- Satter Syed S, Islam MS, Rabbani KS, Talukder MS. Pulsed electromagnetic fields for the treatment of bone fractures. Bangladesh Med Res Counc Bull. 1999;25:6–10. [PubMed]
- Sharrard WJ. A double-blind trial of pulsed electromagnetic fields for delayed union of tibial fractures. J Bone Joint Surg Br. 1990;72:347–354. [PubMed]
- Simonis RB, Parnell EJ, Ray PS, Peacock JL. Electrical treatment of tibial non-union: a prospective, randomized, double-blind trial. Injury. 2003;34:357–362. [PubMed]
- Physiotherapy Evidence Database (PEDro). Available at:http://www.pedro.fhs.usyd.edu.au/index.html. Accessed October 21, 2004.
- Bone growth stimulators: electric and ultrasound. Fallon Community Health Plan, 200311-0002:1-6, 2003. Available at: http://www.fchp.org/. Accessed October 2004.
- Cohen J. Statistical Power Analysis for the Behavioral Sciences. 2nd ed. Hillsdale, NJ: Lawrence Erlbaum; 1988.
- Vincent WJ. Statistics in Kinesiology. Champaign, IL: Human Kinetics; 1995:131.
- Kristiansen TK, Ryaby JP, McCabe J, Frey JJ, Roe LR. Accelerated healing of distal radius fractures with the use of specific, low-intensity ultrasound: a multicenter, prospective, randomized, double-blind, placebo-controlled study. J Bone Joint Surg Am. 1997;79:961–973. [PubMed]
- Rubin C, Bolander M, Ryaby JP, Hadjiargyrou M. The use of low-intensity ultrasound to accelerate the healing of fractures. J Bone Joint Surg Am. 2001;83:259–270. [PubMed]
- Gustilo RB, Anderson JT. Prevention of infection in the treatment of one thousand and twenty-five open fractures of long bones: retrospective and prospective analyses. J Bone Joint Surg Am.1976;58:453–458. [PubMed]
